Clinical & Technical Product Details
Enhancing PVD Treatment Standards
Overview
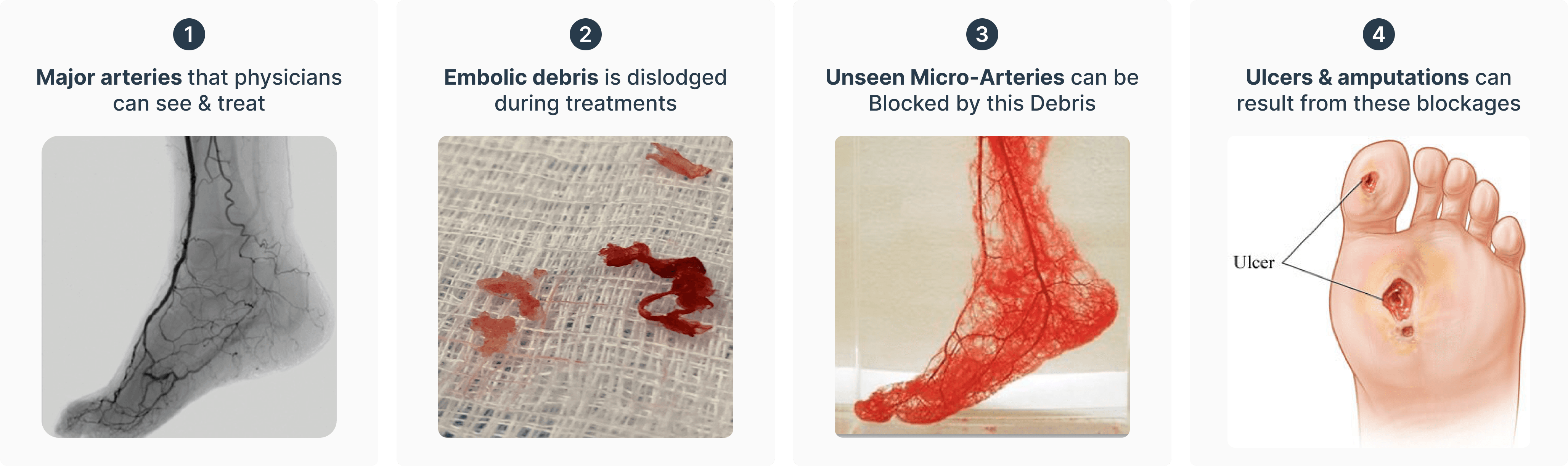
The RoVo System is used in conjunction with standard PVD treatments to remove distal emboli created as blockages are cleared. Using our proprietary Temporary Oclussion Embolectomy (TOE) technique, micro-vascular-systems are protected from the 'downstream' flow of emboli.
Protecting Micro Vascular Systems
PVD treatments such as atherectomies, balloons, and stents are effective options to clear blockages and restore patient blood flow. However, small debris called 'distal emboli' is created in 91%+ of these interventions. The RoVo System allows physicians to safely and effectively remove this debris to help protect the patient's lower vascular system.
Product Highlights
Simultaneous rotation and suction generates a small vortex within patient arteries, stirring up debris for enhanced removal.
Fully FDA approved and available today for use alongside standard PAD treatments (atherectomies, balloons, & stents).
Reimbursable in Outpatient Based Labs (OBLs).
Highly effective and easy to use, with 2MG Medical representatives available to streamline implementation.
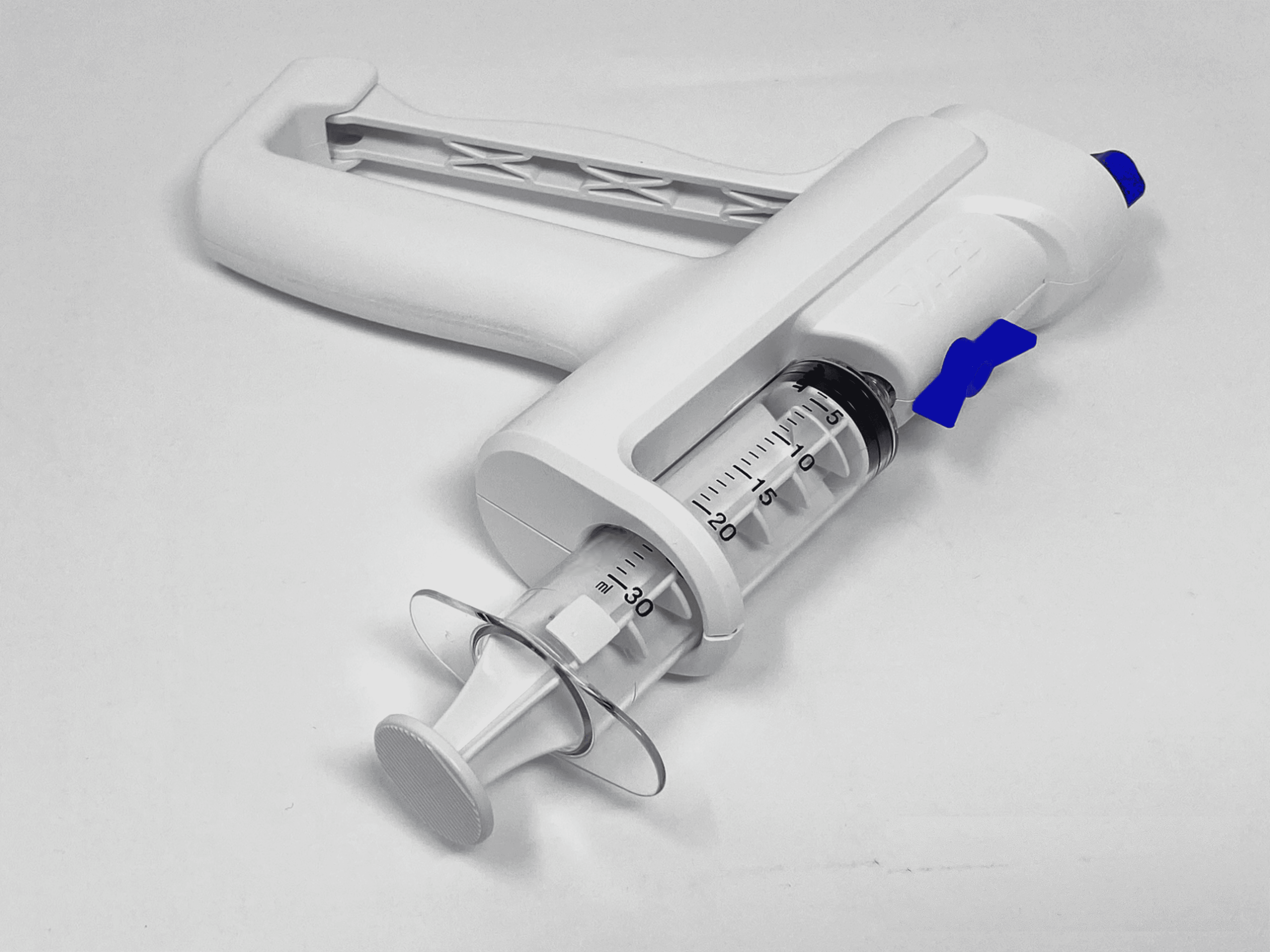
RoVo Handle Sub-Components
Helical Drive Shaft: 2MG's patented helical design rotates the catheter within arteries, creating a small vortex to stir up debris and enhance removal.
Handle: Catheter rotation is driven by squeezing the handle, allowing physicians to retain tactile feedback and adjust rotation frequency for each patient's unique needs.
Syringe: Pulling back the syringe plunger and twisting to lock it in place creates a vacuum, preparing for the suction process to remove debris from patient arteries.
Vacuum Switch: Once the physician is ready, the vacuum switch is engaged to initiate the suction of debris through the catheter and into the syringe.
Catheter: The RoVo System includes a specialized catheter designed to help create vortex.